Our Quality Assurance : Guaranteeing Excellence in Every Product
Quality Assurance Framework
Quality Assurance (QA) is integrated into every stage of production, ensuring products meet stringent international safety and compliance benchmarks. Our approach includes in-process monitoring, batch record reviews, and finished product verification, guaranteeing consistency and reliability across all formulations. By combining advanced testing & detailed documentations.
Quality Assurance at Every Stage of Production
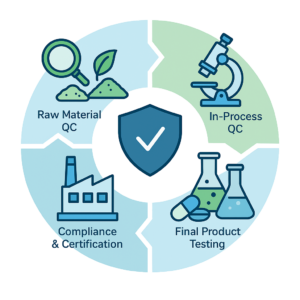
Raw Material Quality Checks
- Supplier Qualification – Only GACP-certified farms and USP/EP-compliant ingredient vendors.
- Identity & Purity Verification – Using HPLC, GC-MS, FTIR, and microscopy.
- Contaminant Control – Heavy metals, pesticides, mycotoxins, and microbial safety testing.
In-Process Quality Monitoring
- Critical Control Point (CCP) Checks – Continuous monitoring during blending, filling, and packing.
- Real-Time Adjustments – Immediate corrective measures to prevent deviations.
- Batch Record Documentation – Detailed logs for traceability and regulatory compliance.
Finished Product Verification
- Potency Confirmation – Ensuring actives meet label claims.
- Stability Testing – Real-time and accelerated conditions to validate shelf life (up to 5 years).
- Packaging Integrity – Leak testing, seal validation, and barrier performance.
Advanced QA Infrastructure
Testing Facilities
- Analytical Labs – Quantification of vitamins, minerals, and botanicals.
- Microbiology Labs – Pathogen detection and preservative efficacy testing.
- Stability Chambers – Controlled environments for shelf life validation.
Digital Quality Management
- ERP-integrated tracking for batch history and documentation.
- Blockchain-enabled ingredient traceability for maximum transparency.
Why LyonsBarry’s QA Facility Stands Out
Quality Assurance Framework
Zero-Defect Philosophy – Every batch undergoes multiple quality checkpoints.
Audit-Ready Operations – Facilities maintained to pass any local or international inspection.
Global Market Expertise – QA processes aligned with 85+ country regulations.
Continuous Improvement – Regular updates to SOPs and technology for better outcomes.












